1
/
of
1
健栄製薬株式会社
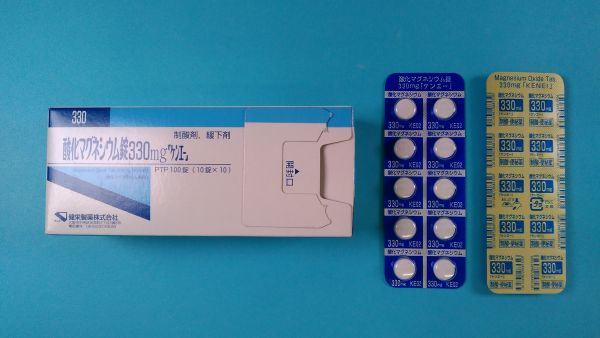
Magnesium Oxide 330mg 100 tablets Magnesium Oxide Magnesium Oxide Constipation Gastritis
Magnesium Oxide 330mg 100 tablets Magnesium Oxide Magnesium Oxide Constipation Gastritis
Regular price
¥1,500 JPY
Regular price
Sale price
¥1,500 JPY
Unit price
/
per
Taxes included.
Couldn't load pickup availability
Magnesium oxide tablets (330mg/tablet) are an oral medication with magnesium oxide as the main ingredient. They are classified as antacids and laxatives, and mainly work by neutralizing stomach acid and regulating intestinal moisture.
I. Basic Drug Information
- Common name : Magnesium oxide
- English name : Magnesium Oxide
- Product Names : Magnesium Oxide Tablets 250mg (Ken-Ai), Magnesium Oxide Tablets 330mg (Ken-Ai), Magnesium Oxide Tablets 500mg (Ken-Ai)
- Dosage form : tablets
-
Indications
- The following conditions can be treated with acid suppression and symptom improvement: gastric and duodenal ulcers, gastritis (including acute and chronic gastritis, and drug-induced gastritis), and upper gastrointestinal dysfunction (including anorexia nervosa, so-called gastroptosis, and hyperacidity).
- constipation
- Prevention of urinary oxalate stones
-
Element
- Magnesium Oxide Tablets 250mg "Ken-Ae" : Each tablet contains 250mg of the active ingredient magnesium oxide. Additives include calcium stearate, crystalline cellulose, croscarmellose sodium, and flavoring.
- Magnesium Oxide Tablets 330mg "Ken-Ae" : Each tablet contains the active ingredient 330mg of magnesium oxide. Additives include calcium stearate, crystalline cellulose, croscarmellose sodium, and flavoring.
- Magnesium Oxide Tablets 500mg "Ken-Ae" : Each tablet contains 500mg of the active ingredient magnesium oxide. Additives include calcium stearate, crystalline cellulose, croscarmellose sodium, and flavoring.
-
Properties
- Magnesium oxide tablets 250mg "Ken-E" : White, round, unbleached tablets with a slightly distinctive aroma. Diameter 8mm, thickness 4.1mm, weight 308mg, identification code: Kama250 KE01, text color: green.
- Magnesium oxide tablets 330mg "Ken-E" : White, round, unbleached tablets with a slightly distinctive aroma. Diameter 9mm, thickness 4.4mm, weight 407mg, identification code: Kama330 KE02, text color: blue.
- Magnesium Oxide Tablets 500mg "Ken-E" : White, round tablets with a slightly distinctive aroma. Diameter 10.5mm, thickness 4.9mm, weight 616mg, identification code is Kama500 KE03, text color is pink.
II. Usage and Dosage
- When used as an antacid : the usual adult dose is 0.5-1.0g of magnesium oxide, taken orally in divided doses.
- When used as a laxative : The usual adult dose is 2g of magnesium oxide, divided into 3 doses, taken orally before or after meals, or once before bedtime.
- When used to prevent the occurrence of calcium oxalate stones in the urinary tract : the usual adult dose is 0.2-0.6g of magnesium oxide per day, taken with plenty of water.
- In either case, the dosage should be adjusted appropriately based on age and symptoms.
III. Taboos
The following patients are prohibited from using this product:
- Patients with hypermagnesemia (may worsen symptoms of hypermagnesemia).
IV. Precautions
-
Special populations
- Patients with cardiac dysfunction: May cause bradycardia and worsen symptoms.
- For patients with diarrhea: May worsen diarrhea.
- Patients with renal dysfunction: are at risk of developing hypermagnesemia.
- Pregnant women or women who may become pregnant: Use only when the benefits of treatment outweigh the risks.
- Breastfeeding women: The benefits of treatment and the nutritional benefits of breast milk should be considered before deciding whether to continue breastfeeding.
- Elderly patients: The dosage should be reduced, and serum magnesium concentration should be monitored regularly. Close observation and caution should be exercised when using the medication, as there are reports of it causing hypermagnesemia and leading to serious consequences.
-
Medication guidance
- This drug may cause hypermagnesemia, especially in patients with constipation. Even with normal renal function or when using doses below the usual level, there have been reports of serious consequences. The following precautions should be taken: Use the minimum necessary dose whenever possible; when administering this drug long-term or to elderly patients, serum magnesium concentrations should be measured regularly; if symptoms such as vomiting, bradycardia, muscle weakness, or drowsiness occur, discontinue use immediately and seek medical attention.
- For medications packaged in PTP, patients should be instructed to remove the PTP sheets before taking the medication to avoid accidental ingestion of PTP sheets, which could lead to esophageal mucosal puncture, perforation, and serious complications such as mediastinitis.
- Long-term, high-dose administration may lead to the formation of stones in the gastrointestinal tract, causing intestinal obstruction.
-
Drug Preservation
- Store at room temperature; avoid moisture after opening.
- The validity period is 3 years.
V. Adverse Reactions
- Allergic reactions : No relevant information was mentioned.
- Digestive system reactions : diarrhea may occur.
-
other
- Major side effects: Hypermagnesemia (frequency unknown), which may cause respiratory depression, altered consciousness, arrhythmia, cardiac arrest, etc. It may also be accompanied by nausea, vomiting, thirst, decreased blood pressure, bradycardia, skin flushing, muscle weakness, drowsiness and other symptoms. Serum magnesium concentration should be monitored.
- Other side effects: Increased serum magnesium levels (frequency unknown).
VI. Drug Interactions
This drug has adsorption and antacid effects, which may affect the absorption and excretion of other drugs. Caution should be exercised when using it in combination with the following drugs:
| Drug category/name | Clinical symptoms and treatment methods | Mechanism/Risk Factors |
|---|---|---|
| Tetracycline antibiotics (tetracycline, minocycline, etc.), quinolone antibiotics (ciprofloxacin, tofloxacin, etc.), bisphosphonates (etidronate sodium, risedronate sodium, etc.), and antiviral drugs (raltegravir, avirbiravir/cobicistat/emtricitabine/tenofovir disoproxil fumarate, etc.). | This may reduce the absorption of these medications and weaken their efficacy; therefore, simultaneous use should be avoided. | Magnesium forms poorly soluble chelates with these drugs, hindering drug absorption. |
| Cefdinir, cefpodoxime proxetil, mycophenolate mofetil, penicillamine | This may reduce the absorption of these medications and weaken their efficacy; therefore, simultaneous use should be avoided. | Mechanism unclear |
| Azithromycin, celecoxib, rosuvastatin, rabeprazole, gabapentin | This may reduce the blood concentration of these drugs. | Mechanism unclear |
| Digitalis preparations (digoxin, digitoxin, etc.), iron supplements, fexofenadine | This may affect the absorption and excretion of these medications; therefore, it is important to take them at intervals. | This may be related to magnesium adsorption or an increase in the pH of the gastrointestinal tract and body fluids. |
| Polycarboxylic Acid | It may weaken the effect of polycarboxylic acid calcium. | Polycarbamide calcium releases calcium to exert its therapeutic effect under acidic conditions. This drug increases the pH in the stomach, thus inhibiting calcium release. |
| Ion exchange resin preparations (calcium polystyrene sulfonate, sodium polystyrene sulfonate) for improving hyperkalemia | This may weaken the effects of these drugs, and there are reports of alkalosis occurring when used together. | This may be due to cation exchange between magnesium and these drugs. |
| Active vitamin D3 preparations (alfacalcidol, calcitriol, etc.) | There is a risk of developing hypermagnesemia. | It may promote the absorption of magnesium in the digestive tract and its reabsorption in the proximal convoluted tubule of the kidney. |
| Active vitamin D3 preparations (alfacalcidol, calcitriol, etc.), large amounts of milk, and calcium supplements. | Milk-alkali syndrome (hypercalcemia, azotemia, alkalosis, etc.) may occur. Close observation is necessary, and medication should be discontinued immediately if symptoms appear. | The mechanism involves persistent metabolic alkalosis leading to increased renal tubular calcium reabsorption; risk factors include hypercalcemia, metabolic alkalosis, and renal dysfunction. |
| LeoCroat | Concomitant use may decrease the blood concentration of riociguat; this drug should be taken 1 hour after riociguat administration. | The increased pH in the digestive tract reduces the bioavailability of liosiegua. |
| Roxadustat, Badassavir | Combining these drugs may weaken their effects. | Mechanism unclear |
| lithium carbonate | There is a risk of developing hypermagnesemia. | Mechanism unclear |
| H2 receptor antagonists (famotidine, ranitidine, lafutidine, etc.) and proton pump inhibitors (omeprazole, lansoprazole, esomeprazole, etc.) | This may weaken the laxative effect of this drug. | The solubility of this drug may be reduced due to an increase in gastric pH. |
| Misoprostol | Combining these medications may increase the risk of diarrhea. | Misoprostol promotes intestinal peristalsis and inhibits the absorption of water and sodium in the small intestine, leading to diarrhea. This drug also has a laxative effect, and combined use may worsen the diarrhea. |
VII. Pharmacological effects
- Moderating biomembrane function : No relevant information was mentioned.
- Improvement of metabolic disorders : No relevant information was mentioned.
- Regulating blood lipids : No relevant information was mentioned.
- Protecting blood vessels : No relevant information was mentioned.
- Other mechanisms of action : This drug has an antacid effect in the stomach and a mild laxative effect in the intestines. Its neutralization reaction with gastric acid is MgO + 2HCl → MgCl₂ + H₂O. The antacid effect does not produce CO₂, making it a non-irritating antacid. 1g of this drug can neutralize approximately 500mL of 0.1mol/L HCl. This drug is insoluble in water, and compared to NaHCO₃, its antacid effect has a slower onset and longer duration. The MgCl₂ produced during neutralization absorbs CO₂, therefore it is often used in combination with NaHCO₃. In the intestines, this drug forms poorly absorbed bicarbonate or carbonate, which absorbs water from the intestinal wall by maintaining osmotic pressure, softening the intestinal contents and thus exerting a mild laxative effect. This drug is not absorbed and does not cause alkalosis.
VIII. Pharmacokinetics
- Absorption : No detailed information was provided regarding absorption.
- Excretion : No specific details regarding excretion are mentioned.
- Other : Pharmacokinetics also includes blood concentration, distribution, metabolism, etc., but the specific details are not mentioned; pharmacokinetic information for specific patient backgrounds, drug interactions, etc. is also not mentioned.
IX. Clinical Research
- Efficacy and safety studies : The antacid effects of magnesium oxide tablets 250mg "Kenya", 330mg "Kenya", and 500mg "Kenya" compared with the standard formulation were confirmed through in vitro Nippon Pharmacopoeia antacid power test and Fuchs modified test; regarding the laxative effect, the bioequivalence of the two was confirmed by comparing the appearance of feces and the incidence of laxative effect after a single oral administration to rats.
- Post-production and sales investigations : No relevant information was mentioned.
10. Packaging Specifications
-
Magnesium oxide tablets 250mg "Ken-E"
- PTP packaging: 100 pieces (10 pieces x 10), 1000 pieces (10 pieces x 100)
- Bulk: 1000 pieces (polyethylene bottle)
-
Magnesium oxide tablets 330mg [Ken-E]
- PTP packaging: 100 pieces (10 pieces x 10), 1000 pieces (10 pieces x 100)
- Bulk: 500 pieces (polyethylene bottle)
-
Magnesium oxide tablets 500mg "Ken-E"
- PTP packaging: 100 pieces (10 pieces x 10), 500 pieces (10 pieces x 50)
- Bulk: 500 pieces (polyethylene bottle)
XI. Production Information
-
manufacturing enterprises
- Manufacturer and Distributor: Kenyi Seed Co., Ltd.
Share