持田製薬株式会社
Epadel Eicosapentaenoic Acid Capsules 300 100 Tablets (Epadel Capsule) - For High Blood Lipids and Lowering Blood Lipids (Ikeda)
Epadel Eicosapentaenoic Acid Capsules 300 100 Tablets (Epadel Capsule) - For High Blood Lipids and Lowering Blood Lipids (Ikeda)
Couldn't load pickup availability
The main ingredient of Epadel Eicosapentaenoic Acid Ethyl Ester Capsules (usually 300mg/capsule) is ethyl eicosapentaenoic acid (EPA ethyl ester), which belongs to the class of unsaturated fatty acid drugs. Its mechanism of action is mainly to inhibit the synthesis of triglycerides and promote their metabolism, thereby reducing the level of triglycerides in the blood. At the same time, it may have certain effects in resisting platelet aggregation and improving endothelial function. It is mainly used clinically to treat hypertriglyceridemia, and can also be used as an adjuvant treatment for cardiovascular diseases such as coronary heart disease to reduce the risk of cardiovascular events.
1. Basic information of the drug
1. Common name : イコサペントエチル
2. English name : EPADEL Capsules 300
3. Product name : Etatoplast 300
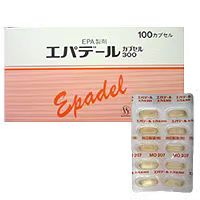
4. Dosage form : soft capsule
5. Ingredients : Each capsule contains the active ingredient イコサペンエチル300mg; additives include イコフェロール in the content, ゼラチン, コハク化ゼラチン in the capsule, etc.
6. Properties : Light yellow transparent soft capsule, PTP logo "MO207".
7. Storage and shelf life : Store at room temperature, shelf life is 3.5 years; avoid high temperature, moisture and light after opening.
2. Indications
1. Improvement of ulcers, pain and cold sensation associated with occlusive arteriosclerosis.
2. Hyperlipidemia.
3. Usage and Dosage
1. Arteriosclerosis obliterans : Adults: 600 mg (2 capsules) each time, 3 times a day, orally immediately after each meal (adjustable according to age and symptoms).
2. Hyperlipidemia : usually 900 mg (3 capsules) twice a day or 600 mg (2 capsules) 3 times a day, taken after meals; if triglyceride is abnormal, the dosage can be increased to 900 mg each time, 3 times a day.
4. Taboos
1. Patients with bleeding (such as hemophilia, gastrointestinal ulcers, urinary tract bleeding, etc.): may cause difficulty in stopping bleeding.
2. Patients who are taking Mifusunプリストン・ミソプロストール: It may aggravate uterine bleeding.
5. Notes
1. Special populations :
a. Pregnant women: Use only when the benefits of treatment outweigh the risks.
b. Breastfeeding women: We need to weigh the pros and cons and decide whether to stop breastfeeding (animal experiments show that the ingredients can enter breast milk).
c. Children: No clinical studies on effectiveness and safety have been conducted.
d. Those with bleeding tendency (menstrual period, before surgery, etc.): The risk of bleeding needs to be closely monitored.
e. Patients with liver/renal dysfunction: Careful monitoring is required (may affect metabolism or excretion).
2. Medication instructions :
a. Do not take on an empty stomach (it affects absorption) and do not chew.
b. The capsule of PTP package must be taken out before taking to avoid accidental swallowing of PTP tablets which may cause damage to the esophagus.
c. Treatment of hyperlipidemia requires diet and exercise therapy first, and control of other cardiovascular risk factors.
d. Conduct blood and blood lipid tests regularly. If the results are ineffective, stop taking the medication and switch to another treatment.
VI. Adverse Reactions
1. Serious side effects :
a. Liver dysfunction, jaundice (elevated AST/ALT, etc.).
b. Atrial fibrillation, atrial flutter (overseas studies show an increased risk at high doses).
2. Other common side effects :
a. Allergic reaction: rash, itching (0.1~5%).
b. Bleeding tendency: subcutaneous bleeding, hematuria, gingival bleeding, etc. (the frequency of some cases is unknown).
c. Digestive system: nausea, abdominal pain, diarrhea, etc. (0.1~5%).
d. Abnormal liver and kidney function indicators (such as increased BUN and creatinine).
7. Drug Interactions
1. Concomitant use is prohibited : Mitsubishi Materia Medica (increases the risk of bleeding).
2. Use with caution : Anticoagulants (such as warfarin), antiplatelet drugs (such as aspirin, ibuprofen): may increase bleeding tendency (additive effect).
8. Pharmacokinetics
1. Absorption: Peak blood concentration is reached 6 hours after taking the drug for a meal, and steady state is reached in about 1 week.
2. Distribution: Mainly distributed in liver, fat, muscle and other tissues, with a high plasma protein binding rate (86.7~98.8%).
3. Metabolism: After deethylation in the small intestine, it is metabolized to acetyl CoA through β-oxidation.
4. Excretion: Mainly excreted through exhalation (44.4%), feces (16.7%), and urine (2.7%).
IX. Clinical Research
1. Occlusive arteriosclerosis : Domestic Phase III trials have shown that the efficacy of improving ulcers and pain is 55.9% (more than effective), and the incidence of side effects is 6.9%.
2. Hyperlipidemia : Phase II/III trials have shown that it can reduce serum total cholesterol (3-6%) and triglycerides (14-20%), and the effect is stable with long-term use; combined with statins, it can reduce cardiovascular events (JELIS study: risk reduction of 19%).
10. Production Information
● Manufacturer and distributor: Mochida Pharmaceutical Co., Ltd.
● Packing: PTP pack (100/500/1000/1050 capsules)
Share